MICROORGANISMS RESPONSIBLE FOR THE SYNTHESIS OF FATTY ACIDS
The microbial population in freshly cut plants differs significantly from that found after the silage fermentation process. This relates to both quantity and taxonomic composition.
Crops that are to be silaged contain both aerobic and anaerobic microorganisms. They directly influence silage quality. Typical groups of microorganisms and their populations in crops used for silage are listed in Table 1. The total number of microorganisms in 1 g of fresh grass can range from 100,000 to 1,000,000,000 CFU [8].
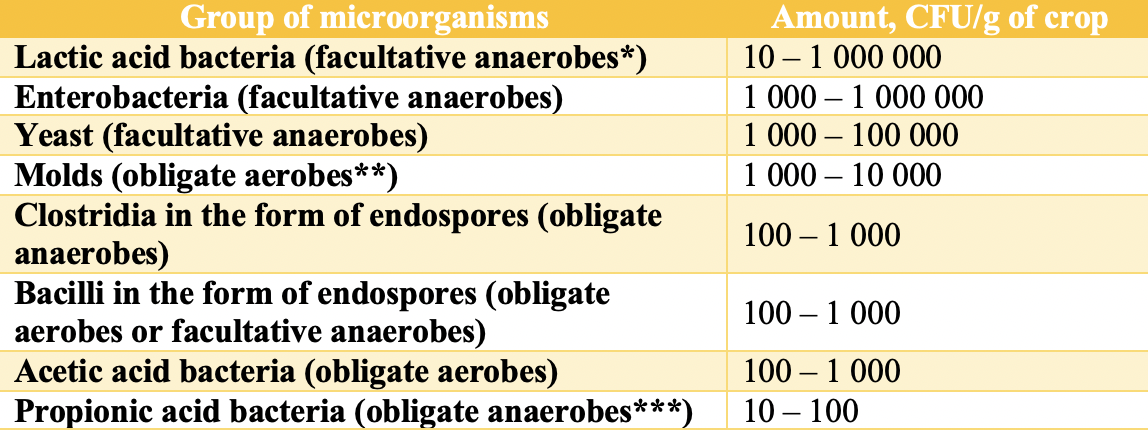
* Facultative anaerobes – can live with or without oxygen.
** Obligate aerobes – require oxygen to survive.
*** Obligate anaerobes – live and grow only in the absence of oxygen.
The dominant populations in plants prior to ensiling are aerobic microorganisms or facultative anaerobes, which require oxygen for their metabolic processes. Lactic acid bacteria are specifically required to initiate fermentation.
The main volatile fatty acids (VFAs) in silage are acetic and butyric acids, and to a lesser extent, propionic acid. We also classify lactic acid as one of the main fatty acids, although it is not volatile.
LACTIC ACID
Lactic acid in silage ensures that fermentation proceeds properly. Its formation contributes to a rapid drop in pH, so a high concentration of this acid is desirable. It also gives the silage a pleasant taste [3].
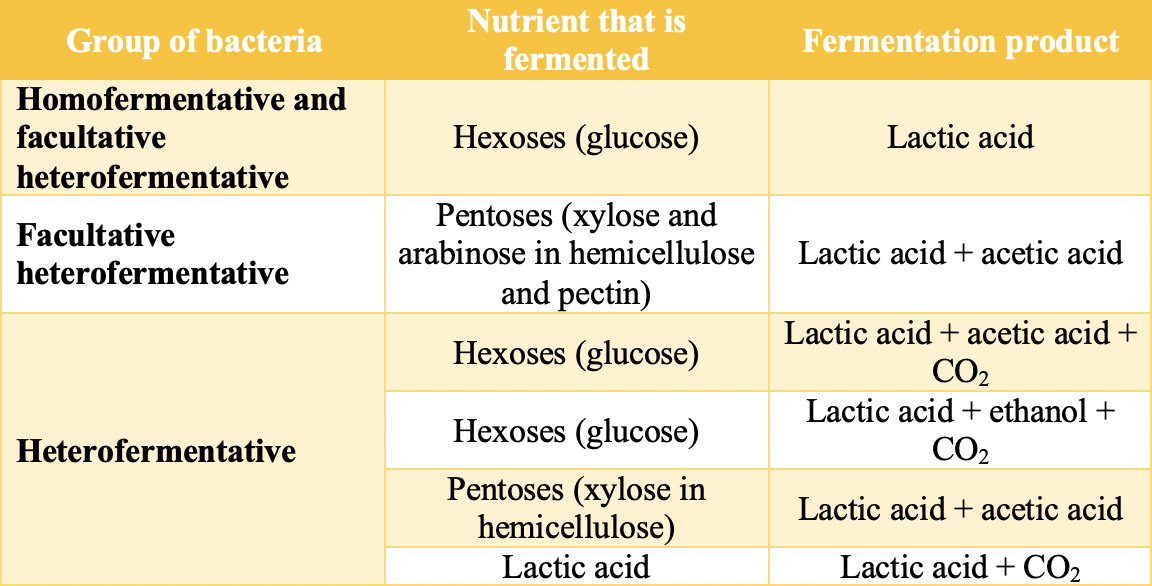
Lactic acid bacteria of the genera Lactobacillus, Pediococcus, Lactococcus, Enterococcus, Streptococcus, and Leuconostoc, which are present in silage, are responsible for the production of lactic acid. All of them produce lactic acid as the main product of sugar fermentation (hexoses and pentoses), but they can also produce other products, including carbon dioxide, acetic acid, and ethanol. Depending on the products of sugar fermentation, bacteria are usually divided into homofermentative (producing only lactic acid) and heterofermentative (producing lactic acid and other fermentation products). Table 2 lists the fermentation products depending on the group of lactic acid bacteria [6,8].
If the lactic acid content in the silage is low, the reasons may be as follows:
- limited fermentation due to high dry matter content (especially grass or legume silage with a dry matter content >50%);
- limited fermentation due to cold weather;
- The sample was taken after a long exposure to aerobic conditions, which led to the breakdown of lactic acid;
- Silage with a high butyric acid content typically has a low lactic acid content [9].
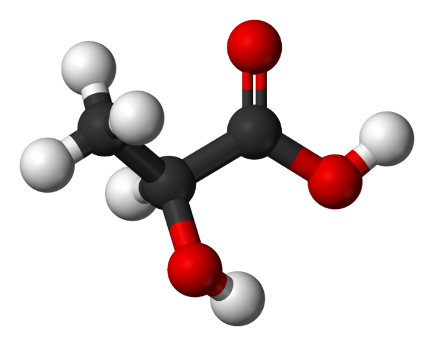
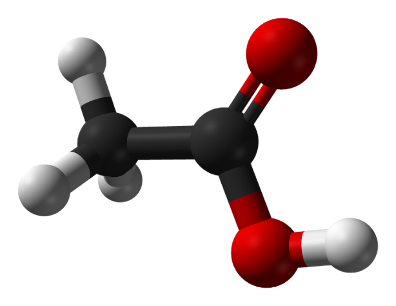
The formula of lactic acid is С3Н6О3, its molecular model is shown in Figure 1.
ACETIC ACID
Acetic acid does not taste very pleasant, but having the right amount of it is essential for proper silage preservation. Silage with low acetic acid content is typically prone to heating [1].
The following factors may contribute to excessively high levels of acetic acid in silage (>3-4% on a dry matter basis):
- high moisture content of the feed (<25% dry matter);
- prolonged fermentation due to high buffer capacity;
- insufficient compaction;
- slow filling of the silo bunker;
- high ammonia levels, as it raises the pH [9].
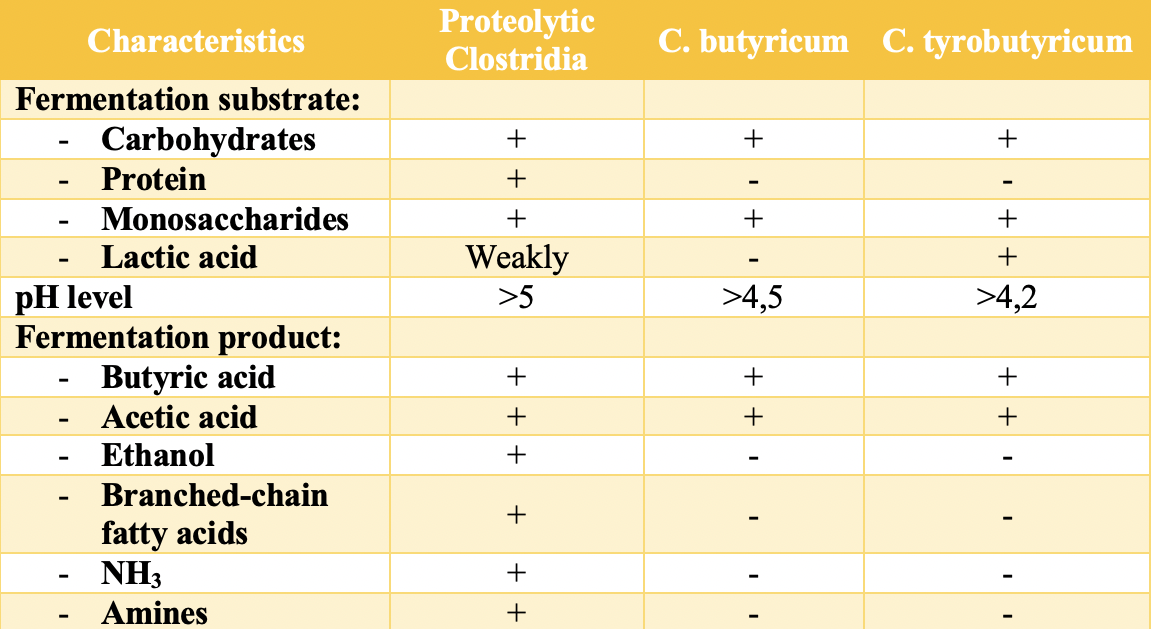
Acetic acid is formed in silages through several pathways. One of these pathways involves the fermentation of sugars by heterofermentative lactic acid bacteria, resulting in the formation of acetic acid alongside lactic acid. In addition, acetic acid can form during secondary fermentation (after the silo is opened and oxygen enters). During the metabolic activity of enterobacteria, which are typically the second most abundant group of microorganisms in silos, glucose is fermented into acetic and formic acids and ethanol. Acetic acid can also be one of the products of undesirable clostridial fermentation (Table 3) [8].
It is important to note that when using inoculants containing the heterofermentative Lactobacillus buchneri, the concentration of acetic acid is higher than normal. Lactobacillus buchneri ferments sugars into both lactic and acetic acids. Another characteristic of this bacteria is that it ferments part of the lactic acid into acetic acid. This helps improve the aerobic stability of silage, as acetic acid is much more effective at inhibiting the growth of yeast and fungi compared to lactic acid. In other words, the production of acetic acid by this bacteria should not be confused with poor fermentation. Feeding such silage to animals has no negative effect on feed intake [7,9].
The formula of acetic acid is СН3СООН, its molecular model is shown in Figure 2.
BUTYRIC ACID
Butyric acid is a volatile fatty acid with a very pungent odor and an unpleasant taste. It is produced during fermentation by butyric acid-producing bacteria of the genus Clostridium at high pH levels (Table 3). The presence of this acid in silage is a sign of feed spoilage [2].
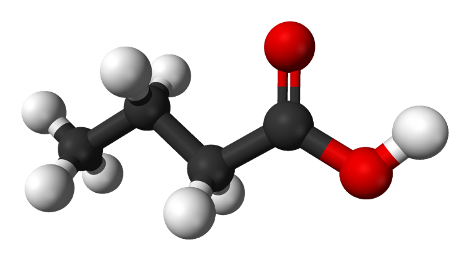
The formula of butyric acid С3Н7СООН, its molecular model is shown in Figure 3.
PROPIONIC ACID
Propionic acid has an unpleasant, sharp, sour odor and taste. It can be produced during the metabolic processes of Propionibacteria. These bacteria ferment both sugars and lactic acid. In addition to propionic acid, this process also produces acetic acid and small amounts of carbon dioxide, formic acid, and isovaleric acid. However, the presence of propionic acid in silage does not necessarily mean that it was produced by propionic acid-producing bacteria. Clostridium propionicum and Selenomonas ruminantium (more characteristic of the rumen) also produce propionic acid as the primary end product of fermentation. Many other microorganisms can also produce small amounts of propionate [4,8].
The role of propionic acid in silages may be to improve aerobic stability (by inhibiting the growth of yeast and mold, similar to how acetic acid does). For this purpose, inoculants containing propionic acid-producing bacteria can be used. However, this is not always a very effective solution, because they produce propionic acid only in silages with slow acidification. Propionibacterium spp. sharply reduce their activity at pH <5.0, so the propionic acid content in silages with normal fermentation is usually low (<0,2–0,3%) .
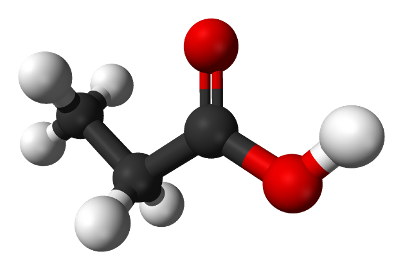
The formula of propionic acid С2Н5СООН, its molecular model is shown in Figure 4.
THE RATIO OF VOLATILE FATTY ACIDS IN SILAGES
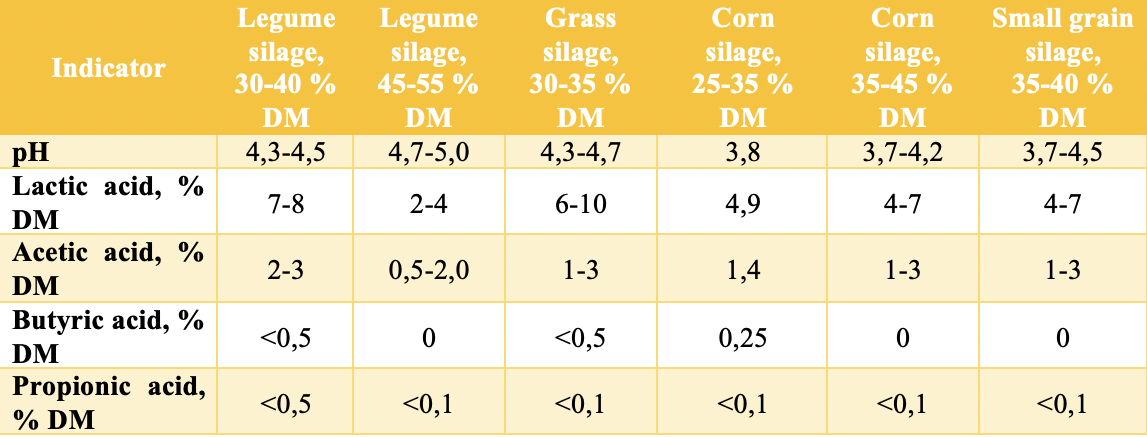
The general recommendation is that the lactic acid content in silage should be approximately 65–70% of the total fatty acids (the higher, the better), and the ratio of lactic acid to acetic acid should not be less than 3:1.
Regarding the fatty acid profiles in different types of silage, various sources may report slightly different values. Table 4 presents data from the University of Wisconsin-Madison and HillLabs [5,9].
REFERENCES
- Eurofins Agro. Acetic acid. https://www.eurofins-agro.com/en/acetic-acid.
- Eurofins Agro. Butyric acid and butyric acid bacteria in silage. https://www.eurofins-agro.com/en/butyric-acid-and-butyric-acid-bacteria.
- Eurofins Agro. Lactic acid. https://www.eurofins-agro.com/en/lactic-acid.
- Eurofins Agro. Propionic acid. https://www.eurofins-agro.com/en/propionic-acid.
- Hill Laboratories. Silage analysis & interpretation: Technical note. R J Hill Laboratories. https://www.hill-labs.co.nz/media/vlsh1tsi/12385v6_technical-note-silage-analysis-interpretation.pdf.
- Muck, R. E. (2010). Silage microbiology and its control through additives. Revista Brasileira de Zootecnia, 39, 183-191. https://doi.org/10.1590/S1516-35982010001300021
- Muck, R. E., Nadeau, E. M. G., McAllister, T. A., Contreras-Govea, F. E., Santos, M. C., & Kung, L. (2018). Silage review: Recent advances and future uses of silage additives. Journal of Dairy Science, 101(5), 3980-4000. https://doi.org/10.3168/jds.2017-13839
- Pahlow, G., & Muck, R. E. (2003). Microbiology of ensiling. In D. R. Buxton, R. E. Muck, & J. H. Harrison (Eds.), Silage Science and Technology (Agronomy Monograph No. 42, pp. 31-93). Madison, WI: American Society of Agronomy. https://doi.org/10.2134/agronmonogr42.c2
- University of Wisconsin-Madison Extension (2016). Fermentation in forage: Understanding silage fermentation https://fyi.extension.wisc.edu/forage/files/2016/10/Fermentation2.pdf.






